Abstract
Background: Chimeric Antigen Receptor (CAR) T cell therapy is arguably one of the most significant breakthroughs in cancer treatment. There are currently five FDA-approved products that are commercially available. However, despite their success, these CAR T-cell therapies cannot induce long-term durable responses in approximately 50% of leukemia or lymphoma-treated patients. Similarly, the results of CAR T-cells in solid tumors have been somewhat disappointing. Therefore, there is an urgent need to design and develop novel CAR T cells with improved efficacy in hematologic malignancies and solid tumors. ROR1 is a carcinoembryonic antigen expressed in different cancers and is associated with tumor stemness, proliferation, metastatic transformation, and treatment resistance. In this project, we optimize an anti-ROR1 CAR using a humanized single-chain variable fragment (scFv) with second (2G) or third-generation (3G) costimulatory domains.
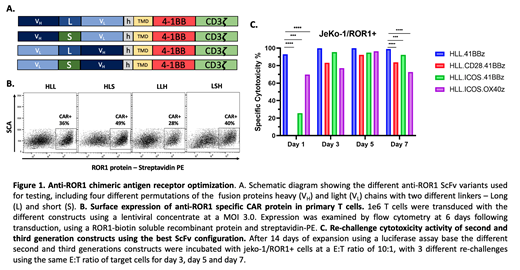
Methods: Several optimization steps in silico were performed using a selected scFv binding domain that targets ROR1. Those included codon optimizations, positional arrangement of heavy-light chains, evaluation of the ideal length of linkers based on tridimensional modeling of the docking between the antibody-like paratope with the target antigen (Figure 1A). After this initial scFv optimization process, we constructed a lentiviral vector that encodes CARs using the selected scFv linked to a transmembrane domain CD28 and different signaling endodomains for 2G and 3G variants (CD28, 41BB, ICOS, OX40), each linked to the T cell receptor CD3z domain. The cytotoxic activity of these constructs was evaluated using an in vitro rechallenge luciferase assay in ROR1 expressing JeKo-1 cells and ROR1(negative) controls.
Results: The 2G 41BB-z construct with V H-V L scFv orientation and a long linker (V H-L-V L) showed optimal cytotoxicity with a CAR expression level in T cells of 36% (Range 28-49% for other constructs, Figure 1B-C). The V H-L-V L 41BB-z construct was evaluated comparatively using a rechallenge cytotoxic assay with 3G constructs that expressed CD28, ICOS, or OX40 signaling domains using JeKo-1 and ROR1(negative) target cells as controls. All the tested constructs showed specific ROR1 medicated cytotoxicity. CD28-41BB-z and ICOS-41BB-z showed the lowest cytotoxicity levels during the Day 1 of the repetitive rechallenge. However, the cytotoxicity levels of those constructs gradually increased during the 7 days of rechallenge and were closed to the levels induced by the 2G- 41BB-z construct (>80% of cytotoxicity). There were no significant differences in CAR T cells subsets generated by the different constructs during the 7 days of rechallenge with a predominance of effector memory phenotype (CCR7-, CD45RA-) and no difference in PD1 expression.
Conclusions: Our results demonstrate that optimization of the CAR constructs enhances T-cell effector function and cytotoxicity against ROR1+ target cells. In previous studies, 3G CARs have shown longer persistence of the transduced T cells in peripheral blood, sustained and regulated cellular activation, improved solid tumor infiltration, and positive modulation of the tumor microenvironment. Our preclinical in vitro optimization demonstrates strategies to generate 3G constructs with a progressive and modulated cytotoxic profile that may confer benefits when tested in vivo in terms of enhanced persistence and lower adverse events profile. Additional experiments in vivo will be presented during the meeting to corroborate our findings.
No relevant conflicts of interest to declare.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal